The Lynes research group is involved in several intertwined projects that relate to interests in the molecular and cellular regulation of the immune response. Central to this interest is their work with metallothionein (MT), a novel stress response protein with an intriguing set of structural features and functions. This small (7 kDa) protein is thiol rich (20 of the 61 amino acids in the consensus sequence are cysteines), which makes the protein an important antioxidant. MT can also capture divalent heavy metal cations, and serves as an important reservoir of essential metals like Cu and Zn, while also protecting other cellular components from the damaging effects of metals like Hg and Cd. The Lynes lab has found that MT has potent immunomodulatory activities that operate in a variety of stressful environments, such as autoimmunity, infection, inflammation, and neoplasia. MT can suppress antibody production, and injected anti-MT monoclonal antibody can enhance a normal immune response (done in collaboration with Dr Lawrence Silbart). Recently, the Lynes lab group has collaborated with the David Knecht lab to show that MT can act as a chemokine: a soluble signal that can initiate directional movement of cells. This suggests that one of the ways that MT may influence immune capacity is by directing the trafficking of cells within the body. Under stress, the normal gradients of MT may be disturbed by overlying and inappropriate gradients, thus diminishing immune efficacy. Our current studies are focused on the characterization of the molecular interactions of MT with the cell, and the signal transduction cascades that are influenced by these interactions.
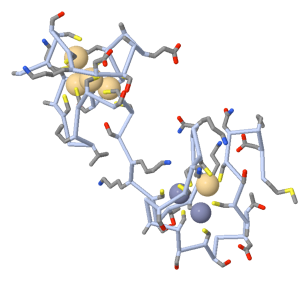
Metallothionein Structure
A second line of research in the Lynes laboratory relates to the development of new technologies for the study of the immune response and other biological systems. Again, in collaboration with Dr. Knecht, and with a small biotechnology company in New York (Applied Biophysics, Inc.) we have developed a microelectrode system for the measurement of cellular movement in a chemotactic gradient. As cells respond to the gradient, they move across a microelectrode that has been photolithographed on the substrate. Cell arrival at this miniature speed bump can be measured in real-time by the instrumentation, allowing a rapid and objective assessment of cell movement. The system is sensitive enough to determine the arrival of a single cell at the target, and can quantify the magnitude and speed of the responding population. This technology is useful for the assessment of intrinsic cellular responses to chemotactic signals, as well as for the determination of what effects drugs will have on that response. Current plans are to adapt this system to studies of metastatic cell movement.
A third line of research in the Lynes laboratory relies on a physical phenomenon called surface plasmon resonance (SPR) to detect molecular interactions at a gold/dielectric interface. In SPR, capture of analyte by a molecule immobilized on a gold surface results in an increase in the refractive index at the surface. This increase can be measured by determining the angle at which light will transfer some of its energy to the gold surface before and after capture of the analyte. The angle increase is proportional to the amount of analyte captured. One drawback of the established SPR technologies is a practical limitation on the number of simultaneous measurements that can be done. At present that limit is usually four independent measurements. In collaboration with the staff at a small biotechnology company in East Hartford (Ciencia, Inc.), the Lynes laboratory has developed ways to take advantage of a different optical configuration (Grating-Coupled Surface Plasmon Resonance Imaging, or GCSPRI) that allows multiplexing of the SPR measurement. At present, we can do up to 1,000 independent and simultaneous assays in the same 10 ul volume. Since the system does not rely on labeled reagents, it is versatile and provides a direct assay of the molecular binding events, both in absolute amounts as well as in terms of binding and dissociation kinetics. Our current work is aimed at developing applications for real-time cytometry, pathogen detection, and toxin detection.

A photo of a GCSPRI Instrument. The gold-coated chip with its microarray of capture proteins is loaded into the flow cell. Then, samples are pumped over the chip with the peristaltic pump and binding is observed with the CCD camera